
IV compounding for hazardous and nonhazardous drugs comes with challenges. These challenges can affect the safety of both patients and pharmacy staff. The Compounder is designed to meet these challenges. What’s more, it does so while enhancing the efficiency of your compounding operations. The Compounder family of automated compounding systems protects the operator from exposure to (hazardous) chemicals. By preparing IV admixtures virtually hands-free, this technology provides a consistent, safe and fully controlled process. It’s faster, safer and more accurate than manual compounding.

The Compounder 5
Ideal for smaller cabinets or for installation in the ward’s medication room. 5 products prepared in 1 production run. Room for 12 inputs (vials, or other containers) and 4 transport syringes. Maximum 4 API’s per production run.

The Compounder 8
Ideal for mid-size cabinets and preparing weekly medication regimes like OPAT. 8 products prepared in 1 production run. Room for 18 inputs (vials, or other containers) and 6 transport syringes. Maximum 6 API’s per production run.

The Compounder 10
Ideal for mid-size cabinets and preparing weekly medication regimes like OPAT. 10 products prepared in 1 production run. Room for 24 inputs (vials, or other containers) and 8 transport syringes. Maximum 8 API’s per production run.
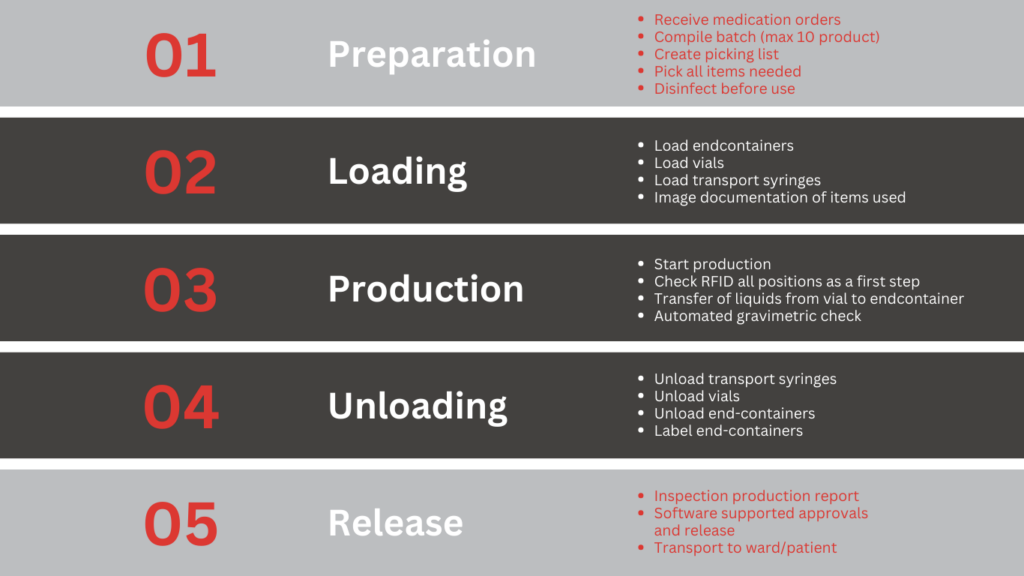
Production process
The Compounder is usually installed in a LAF cabinet or a hood, under a plenum or sometimes even on a desk in a medication room on a ward. Pharmacy staff receives medication orders through file transfer or HL7 integration with an HIS or PIS and plans production runs of maximum 10 orders. The planned batch cannot exceed 8 API’s. Items needed are picked and delivered to the cleanroom. Before starting a production batch vials and bags need to be spiked and coupled to a Compounder adapter, next to The Compounder or in an adjoining cabinet. After the guided loading process, the operator starts the production and can focus on other tasks (mostly preparing the next production run, reconstituting powders) until production is finished. Unloading the finished products is done one by one, a label is printed and the products can be transferred to the release procedure.

Main parts of The Compounder
- Control cabinet: “management” or “brains” of The Compounder.
- Output plate: room for 10 end-containers, each on an individual scale and 2 waste positions.
- Input plate: room for 24 spiked vials.
- Adapters: both inputs and outputs are put on the compounder with an adapter.
- Vision unit: takes pictures of products during loading and unloading, links products to an RFID chip, identification of products.
- HMI unit: screen that guides operator through process.
- Syringe racks: 4 doors that hold 8 transport syringes to transfer liquids (8 syringes = 8 API’s maximum per production batch).

The adapters
Freedom to operate. Freedom to use the vial spikes, bagspikes, and end-containers you want. That is what The Compounder offers for automating your compounding process. Most major brands of Luer-Lock connectors and CSTD’s can be used on The Compounder. That is achieved by our customizable adapters. The Compounder adapters will be made on order, for your preferred vial spike, bag spike or CSTD. We also customize the colour of the adapters as a supporting factor for pharmacy staff. There is a vast amount of disposables and end-containers available worldwide, therefore we cannot guarantee 100% coverage. If a consumable or end-container presents a physical shape or another parameter that limits the use on The Compounder we can advise on the best alternative.
